Determine the Molecular Geometry of ClF3: A Comprehensive Guide
Understanding molecular geometry is essential in chemistry, as it helps predict the shape and properties of molecules. Determining the molecular geometry of ClF3 (Chlorine trifluoride) is a fascinating topic that combines theoretical knowledge with practical applications. This article will delve into the intricacies of ClF3's molecular geometry, providing insights that are both educational and applicable.
Chemistry enthusiasts and students often encounter challenges when trying to understand the molecular geometry of compounds like ClF3. This article aims to simplify the process by breaking down complex concepts into digestible information. By the end of this guide, you will have a clear understanding of how to determine the molecular geometry of ClF3.
Whether you're studying for exams, working on a research project, or simply curious about molecular structures, this article will serve as a valuable resource. We will explore the VSEPR theory, Lewis structures, and other essential principles that govern molecular geometry. Let's dive in!
Introduction to ClF3
Chlorine trifluoride (ClF3) is a fascinating molecule that plays a crucial role in various chemical processes. It is a colorless, toxic, and highly reactive gas that is used in industries such as semiconductor manufacturing and rocket propellants. To understand its reactivity and properties, we must first explore its molecular geometry.
What is ClF3?
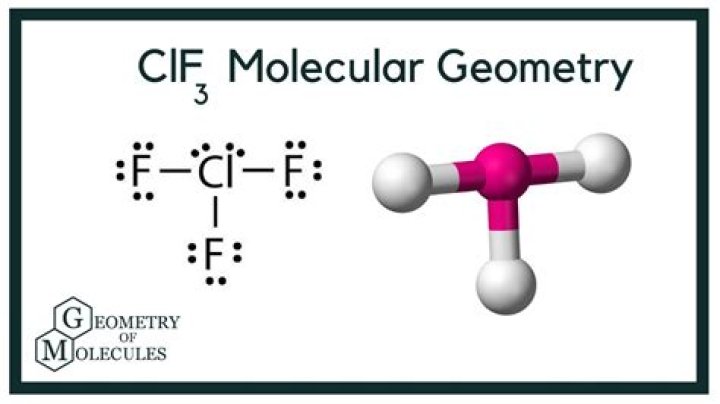
ClF3 consists of one chlorine atom bonded to three fluorine atoms. Its molecular formula indicates that it has a central chlorine atom surrounded by three fluorine atoms and two lone pairs of electrons. This arrangement gives ClF3 its unique molecular geometry.
Why is Molecular Geometry Important?
Molecular geometry determines the spatial arrangement of atoms in a molecule, which influences its chemical properties, reactivity, and physical characteristics. By understanding the molecular geometry of ClF3, we can predict how it interacts with other molecules and its behavior in different environments.
VSEPR Theory and Its Role in Determining Molecular Geometry
The Valence Shell Electron Pair Repulsion (VSEPR) theory is a cornerstone of molecular geometry. It explains how electron pairs around a central atom repel each other, leading to specific molecular shapes. In the case of ClF3, VSEPR theory helps us determine its molecular geometry by considering the repulsion between bonding pairs and lone pairs of electrons.
Key Principles of VSEPR Theory
- Electron pairs around a central atom repel each other, causing them to arrange themselves as far apart as possible.
- Bonding pairs and lone pairs of electrons contribute to the overall shape of the molecule.
- Lone pairs occupy more space than bonding pairs, leading to distortions in molecular geometry.
Understanding the Lewis Structure of ClF3
The Lewis structure of ClF3 provides a visual representation of its electron distribution. It shows how the chlorine atom forms single bonds with three fluorine atoms and has two lone pairs of electrons. This structure is essential for determining the molecular geometry of ClF3.
Steps to Draw the Lewis Structure of ClF3
- Determine the total number of valence electrons: Chlorine contributes 7 electrons, and each fluorine atom contributes 7 electrons, resulting in a total of 28 valence electrons.
- Place the chlorine atom in the center and surround it with three fluorine atoms.
- Form single bonds between chlorine and fluorine atoms.
- Complete the octets of the fluorine atoms and place the remaining electrons as lone pairs on the chlorine atom.
Determining the Molecular Shape of ClF3
Based on the VSEPR theory and Lewis structure, ClF3 has a T-shaped molecular geometry. The two lone pairs of electrons on the chlorine atom cause significant repulsion, pushing the three fluorine atoms into a T-shaped arrangement.
Factors Influencing Molecular Shape
- Number of bonding pairs and lone pairs of electrons.
- Electron pair repulsion and its effect on bond angles.
- Stability of the molecule in its lowest energy state.
Analyzing Bond Angles in ClF3
The bond angles in ClF3 are approximately 90° and 180°. These angles result from the repulsion between the lone pairs and bonding pairs of electrons. The lone pairs occupy more space, causing the bond angles to deviate slightly from the ideal angles predicted by VSEPR theory.
Significance of Bond Angles
Bond angles are critical in determining the shape and properties of a molecule. In ClF3, the T-shaped geometry and bond angles influence its reactivity and interactions with other molecules.
Electronic Geometry vs Molecular Geometry
While the electronic geometry of ClF3 is trigonal bipyramidal, its molecular geometry is T-shaped. The difference arises due to the presence of lone pairs, which affect the spatial arrangement of atoms in the molecule.
Key Differences
- Electronic geometry considers all electron pairs, including lone pairs.
- Molecular geometry focuses only on the positions of atoms in the molecule.
Dipole Moment and Polarity of ClF3
ClF3 is a polar molecule due to its asymmetrical shape and the difference in electronegativity between chlorine and fluorine atoms. The dipole moments of the Cl-F bonds do not cancel out, resulting in a net dipole moment for the molecule.
Factors Affecting Polarity
- Electronegativity difference between bonded atoms.
- Molecular geometry and symmetry.
- Distribution of electron density in the molecule.
Applications of ClF3 in Chemistry
ClF3 finds applications in various fields, including semiconductor manufacturing, rocket propellants, and chemical synthesis. Its unique molecular geometry and reactivity make it an essential compound in industrial processes.
Industrial Uses
- Etching silicon wafers in semiconductor production.
- Oxidizing agent in rocket fuels.
- Synthesizing other chlorine and fluorine compounds.
Common Mistakes to Avoid When Determining Molecular Geometry
Students and researchers often make errors when determining the molecular geometry of ClF3. Some common mistakes include:
- Ignoring the effect of lone pairs on molecular shape.
- Misapplying VSEPR theory to predict bond angles.
- Confusing electronic geometry with molecular geometry.
Conclusion and Final Thoughts
In conclusion, determining the molecular geometry of ClF3 involves understanding VSEPR theory, Lewis structures, and the role of lone pairs in shaping the molecule. ClF3's T-shaped geometry, bond angles, and dipole moment contribute to its unique properties and applications in chemistry.
We encourage readers to explore further resources and practice applying these concepts to other molecules. Share your thoughts and questions in the comments section below. For more insightful articles on chemistry and molecular geometry, explore our website and stay updated on the latest developments in the field!
References:
- https://www.chemguide.co.uk/inorganic/period3/chlorides.html
- https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Modules_(Physical_and_Theoretical_Chemistry)/Chemical_Bonding/Fundamentals_of_Chemical_Bonding/Valence_Shell_Electron_Pair_Repulsion_Theory


