Is Linear CO2 Polar or Nonpolar? A Comprehensive Guide
Carbon dioxide (CO2) is one of the most abundant molecules on Earth, playing a critical role in various natural processes such as photosynthesis, respiration, and the carbon cycle. However, understanding its molecular properties, particularly whether it is polar or nonpolar, is essential for students, researchers, and professionals in chemistry, environmental science, and engineering. In this article, we will delve into the fascinating world of CO2's molecular structure and its polarity, providing a thorough analysis to help you understand this topic in depth.
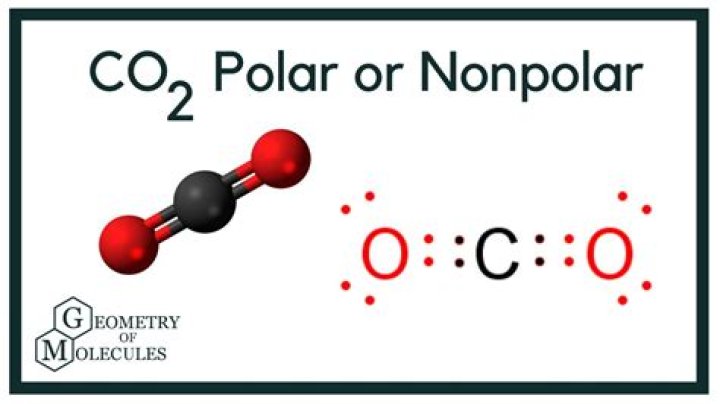
CO2 is a linear molecule consisting of one carbon atom double-bonded to two oxygen atoms. Its unique geometry and electronic properties make it an interesting subject of study in chemistry. Whether CO2 is polar or nonpolar depends on its molecular structure and the distribution of electrical charges within the molecule.
This article aims to provide a comprehensive explanation of CO2's polarity, including its molecular geometry, bond polarity, and dipole moments. By the end of this article, you will have a clear understanding of why CO2 is classified as either polar or nonpolar and how this classification impacts its behavior in various chemical and physical processes.
Introduction to CO2
Carbon dioxide (CO2) is a naturally occurring molecule that plays a crucial role in maintaining life on Earth. It is a colorless, odorless gas that exists in the atmosphere and is essential for plant growth through photosynthesis. CO2 is also a greenhouse gas, contributing significantly to global warming and climate change.
From a chemical perspective, CO2 is classified as a diatomic molecule with a linear structure. The molecule consists of one carbon atom bonded to two oxygen atoms via double bonds. This arrangement gives CO2 its distinct properties, including its polarity or nonpolarity.
Chemical Formula and Structure
The chemical formula of CO2 is straightforward: one carbon atom (C) and two oxygen atoms (O). The carbon atom forms double covalent bonds with each oxygen atom, resulting in a linear geometry. This linear shape is critical in determining whether CO2 is polar or nonpolar.
Molecular Structure of CO2
The molecular structure of CO2 is a critical factor in determining its polarity. CO2 has a linear geometry, meaning the carbon atom is positioned symmetrically between the two oxygen atoms. This symmetry plays a significant role in the distribution of electrical charges within the molecule.
Bond Angle and Geometry
In CO2, the bond angle between the carbon and oxygen atoms is 180 degrees. This perfect linear arrangement ensures that the molecule is symmetric, which affects its overall polarity. The symmetry of CO2's structure is a key factor in understanding why it is classified as nonpolar.
Bond Polarity in CO2
Although the CO2 molecule as a whole is nonpolar, the individual bonds between carbon and oxygen are polar. Oxygen is more electronegative than carbon, meaning it attracts the shared electrons in the covalent bonds more strongly. This creates a partial negative charge on the oxygen atoms and a partial positive charge on the carbon atom.
However, due to the linear geometry of CO2, the dipole moments of the two C=O bonds cancel each other out. This cancellation results in a net dipole moment of zero, making the molecule nonpolar.
Electronegativity Differences
- Oxygen has an electronegativity value of 3.44.
- Carbon has an electronegativity value of 2.55.
- The difference in electronegativity creates a polar bond, but the symmetry of the molecule negates the overall polarity.
Dipole Moment in CO2
The dipole moment is a measure of the separation of positive and negative charges within a molecule. In CO2, each C=O bond has a dipole moment due to the electronegativity difference between carbon and oxygen. However, because the molecule is linear and symmetric, the dipole moments of the two bonds cancel each other out.
This cancellation results in a net dipole moment of zero, confirming that CO2 is a nonpolar molecule despite having polar bonds.
Measuring Dipole Moments
Dipole moments are typically measured in debyes (D). For CO2, the dipole moment is zero, which is consistent with its nonpolar classification. This measurement is supported by experimental data and theoretical calculations.
Is CO2 Polar or Nonpolar?
Based on the molecular structure and dipole moment analysis, CO2 is classified as a nonpolar molecule. Although the individual C=O bonds are polar, the symmetry of the molecule ensures that the dipole moments cancel out, resulting in no net polarity.
This classification is important in understanding how CO2 interacts with other molecules and its behavior in various chemical and physical processes.
Practical Implications
- CO2's nonpolar nature makes it soluble in nonpolar solvents like hexane.
- It is insoluble in polar solvents like water, except under high pressure.
- This property is utilized in carbonated beverages and other industrial applications.
Factors Affecting CO2 Polarity
Several factors contribute to the polarity or nonpolarity of CO2:
Molecular Symmetry
The linear and symmetric structure of CO2 is the primary factor determining its nonpolar nature. Any deviation from this symmetry, such as in bent molecules like water (H2O), would result in a polar molecule.
Electronegativity Differences
While electronegativity differences create polar bonds, the symmetry of CO2 ensures that these polarities cancel out, resulting in a nonpolar molecule.
Real-World Applications of CO2 Polarity
The nonpolar nature of CO2 has significant implications in various fields:
Industrial Applications
- CO2 is used as a refrigerant in cooling systems due to its nonpolar properties.
- It is employed in fire extinguishers because it does not react with most materials.
- CO2 is used in enhanced oil recovery processes to extract oil from underground reservoirs.
Environmental Impact
Understanding CO2's polarity is crucial in studying its role in the greenhouse effect and climate change. Its nonpolar nature affects how it interacts with other atmospheric gases and particles.
Common Misconceptions About CO2 Polarity
There are several misconceptions about CO2's polarity that need clarification:
Confusion with Water
Some people mistakenly believe that CO2 is polar because water (H2O) is polar. However, the difference lies in molecular geometry: water is bent, while CO2 is linear.
Misinterpretation of Electronegativity
Although CO2 has polar bonds due to electronegativity differences, its symmetry ensures that the molecule as a whole is nonpolar.
Comparison with Other Molecules
To better understand CO2's polarity, it is helpful to compare it with other molecules:
Comparison with Water
Water (H2O) is a polar molecule due to its bent geometry, which prevents the dipole moments from canceling out. In contrast, CO2's linear geometry ensures that its dipole moments cancel, making it nonpolar.
Comparison with Methane
Methane (CH4) is another nonpolar molecule with a tetrahedral geometry. Like CO2, its symmetry results in no net dipole moment, despite having polar bonds.
Conclusion and Next Steps
In conclusion, CO2 is a nonpolar molecule due to its linear geometry and symmetric distribution of electrical charges. Although it has polar bonds, the dipole moments cancel out, resulting in no net polarity. Understanding CO2's polarity is essential for various applications in chemistry, industry, and environmental science.
We encourage you to explore further topics related to molecular polarity, such as the polarity of other gases and their applications. Share this article with your peers and leave a comment below if you have any questions or insights. Together, we can deepen our understanding of the fascinating world of chemistry.



