Methane Lewis Dot: Understanding the Chemistry Behind the Dot Structure
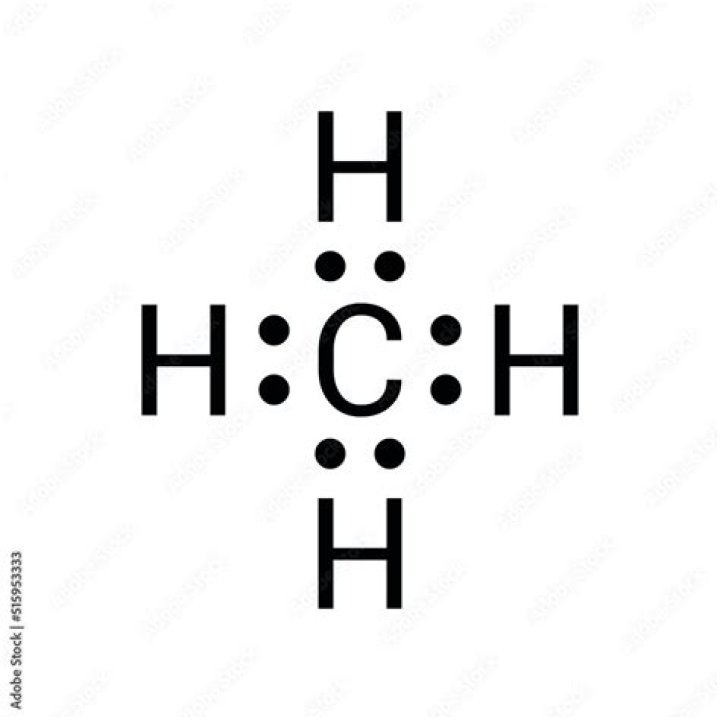
Understanding the Methane Lewis Dot structure is fundamental for anyone studying chemistry. It represents the arrangement of valence electrons around atoms and provides insight into molecular bonding. Whether you're a student, researcher, or simply curious about the basics of chemistry, this guide will provide a comprehensive overview of methane's Lewis dot structure and its significance.
Methane, one of the simplest hydrocarbons, plays a crucial role in organic chemistry and environmental science. Its molecular formula, CH₄, indicates one carbon atom bonded to four hydrogen atoms. The Lewis dot structure helps visualize how these atoms bond, making it easier to understand methane's properties and behavior.
In this article, we'll delve into the intricacies of the Methane Lewis Dot structure, explore its applications, and highlight its importance in chemistry. Whether you're new to the subject or looking to deepen your knowledge, this guide will cover everything you need to know about methane's electron distribution and molecular structure.
What is Methane Lewis Dot Structure?
The Methane Lewis Dot structure is a graphical representation that shows how valence electrons are distributed among atoms in a molecule. In the case of methane (CH₄), the structure illustrates the single covalent bonds between one carbon atom and four hydrogen atoms. Each bond consists of two shared electrons, ensuring that all atoms achieve a stable electron configuration.
Methane's Lewis dot structure is relatively straightforward, as carbon contributes four valence electrons, while each hydrogen atom contributes one. Together, they form a tetrahedral shape, which is essential for understanding methane's geometry and reactivity.
Why Study Methane Lewis Dot?
- To understand molecular bonding and electron distribution.
- To predict the shape and properties of methane molecules.
- To apply this knowledge in fields like organic chemistry and environmental science.
Understanding Methane's Chemical Composition
Methane (CH₄) is composed of one carbon atom and four hydrogen atoms. Carbon, located in Group 14 of the periodic table, has four valence electrons. Hydrogen, in Group 1, contributes one valence electron per atom. Together, they form a stable molecule through covalent bonding, where electrons are shared equally between atoms.
The stability of methane arises from the octet rule, which states that atoms tend to gain, lose, or share electrons to achieve a full outer shell of eight electrons. In methane's case, carbon achieves this configuration by forming four covalent bonds with hydrogen atoms.
Key Features of Methane
- Molecular formula: CH₄
- Valence electrons: 8 (4 from carbon + 1 from each hydrogen)
- Bond type: Covalent
- Molecular geometry: Tetrahedral
Steps to Draw Methane Lewis Dot Structure
Drawing the Methane Lewis Dot structure involves a systematic approach. Follow these steps to accurately represent the molecule:
- Determine the total number of valence electrons: Carbon contributes 4, and each hydrogen contributes 1, totaling 8 electrons.
- Place the carbon atom at the center and surround it with four hydrogen atoms.
- Form single covalent bonds between carbon and each hydrogen atom by sharing two electrons per bond.
- Ensure all atoms have a stable electron configuration, with carbon achieving an octet and each hydrogen having two electrons.
Common Mistakes to Avoid
- Forgetting to include all valence electrons.
- Incorrect placement of atoms in the structure.
- Overlooking the importance of electron sharing in covalent bonds.
The Role of Methane in Organic Chemistry
Methane serves as the foundation for organic chemistry, being the simplest alkane and a primary component of natural gas. Its stability and reactivity make it an essential molecule in various chemical reactions, such as combustion and substitution reactions.
In organic chemistry, methane's Lewis dot structure helps explain its behavior in different environments. For instance, methane's tetrahedral geometry influences its interactions with other molecules and its role in energy production.
Applications of Methane
- Energy source: Methane is a key component of natural gas, widely used for heating and electricity generation.
- Industrial applications: It serves as a raw material for producing hydrogen, methanol, and other chemicals.
- Environmental impact: Methane is a potent greenhouse gas, contributing significantly to global warming.
Methane's Molecular Geometry and Bond Angles
The Methane Lewis Dot structure reveals methane's tetrahedral molecular geometry, where the carbon atom is at the center, and the four hydrogen atoms are equidistant from one another. This arrangement results in bond angles of approximately 109.5°, which is characteristic of sp³ hybridized orbitals.
Understanding methane's geometry is vital for predicting its physical and chemical properties, such as its boiling point, solubility, and reactivity with other substances.
Factors Influencing Methane's Geometry
- Electron repulsion: The repulsion between electron pairs determines the molecule's shape.
- Hybridization: Carbon's sp³ hybridization leads to the tetrahedral arrangement.
- Environmental factors: External conditions can slightly affect bond angles and geometry.
Comparing Methane with Other Hydrocarbons
While methane is the simplest hydrocarbon, comparing it with other molecules like ethane (C₂H₆) and propane (C₃H₈) highlights the importance of Lewis dot structures in understanding molecular bonding. Each hydrocarbon has a unique structure and properties, influenced by the number of carbon atoms and their bonding patterns.
For example, ethane's Lewis dot structure shows two carbon atoms connected by a single bond, with each carbon also bonded to three hydrogen atoms. This difference in structure results in variations in reactivity and physical properties.
Key Differences Between Methane and Other Hydrocarbons
- Number of carbon atoms: Methane has one, while ethane and propane have two and three, respectively.
- Bonding patterns: Methane exhibits simple tetrahedral geometry, while larger hydrocarbons have more complex structures.
- Reactivity: Methane is less reactive compared to larger hydrocarbons due to its simpler structure.
Environmental Impact of Methane
Methane is a significant greenhouse gas, with a global warming potential 28 times greater than carbon dioxide over a 100-year period. Its release into the atmosphere contributes to climate change, making it a critical focus for environmental scientists and policymakers.
Understanding methane's Lewis dot structure and molecular properties helps researchers develop strategies to mitigate its environmental impact. For example, capturing methane emissions from landfills and agricultural activities can reduce its contribution to global warming.
Efforts to Reduce Methane Emissions
- Improved agricultural practices: Reducing methane emissions from livestock and rice cultivation.
- Landfill management: Capturing methane from decomposing organic waste.
- Energy efficiency: Reducing methane leaks during natural gas production and transportation.
Conclusion
In conclusion, the Methane Lewis Dot structure provides a fundamental understanding of methane's molecular bonding and geometry. By studying this structure, we gain insights into methane's properties, applications, and environmental impact. Whether you're exploring organic chemistry or addressing climate change, methane's Lewis dot structure serves as a vital tool for scientific discovery.
We encourage readers to share their thoughts and questions in the comments section below. For further reading, explore related topics such as ethane and propane Lewis dot structures, or delve into the broader implications of methane in environmental science. Together, let's deepen our understanding of the world around us.



