Understanding Carbon Dioxide Molecular Shape: A Comprehensive Guide
Carbon dioxide molecular shape is a fundamental concept in chemistry that plays a crucial role in understanding the behavior and properties of this vital molecule. As one of the most abundant gases in Earth's atmosphere, carbon dioxide (CO2) has significant implications for both environmental science and industrial applications. In this article, we will delve into the molecular geometry of carbon dioxide, exploring its structure, properties, and significance in various fields.
Carbon dioxide is a naturally occurring gas that exists in our atmosphere and plays a critical role in maintaining the Earth's climate. It is also an essential component of many biological processes, such as photosynthesis and respiration. Understanding its molecular shape helps scientists predict its reactivity, stability, and interactions with other molecules.
This article aims to provide a detailed and comprehensive overview of the carbon dioxide molecular shape. Whether you're a student, researcher, or simply someone interested in chemistry, this guide will cover everything you need to know about the structure and properties of CO2, supported by scientific data and reliable sources.
Introduction to Carbon Dioxide
What is Carbon Dioxide?
Carbon dioxide is a chemical compound consisting of one carbon atom covalently bonded to two oxygen atoms. Its molecular formula is CO2, and it is a colorless, odorless gas under standard conditions. CO2 is produced through various natural processes, including respiration, decomposition, and volcanic activity. It is also emitted during human activities such as fossil fuel combustion and industrial processes.
Why is Carbon Dioxide Important?
Carbon dioxide is a critical molecule in Earth's atmosphere, playing a significant role in the greenhouse effect, which helps regulate the planet's temperature. Additionally, CO2 is an essential component of photosynthesis, the process by which plants convert sunlight into energy. Its importance extends to industrial applications, including carbonated beverages, fire extinguishers, and refrigeration systems.
What is Molecular Shape?
Molecular shape refers to the three-dimensional arrangement of atoms in a molecule. This geometric configuration determines the molecule's physical and chemical properties, such as its reactivity, polarity, and boiling point. Understanding molecular shape requires knowledge of valence shell electron pair repulsion (VSEPR) theory, which predicts the geometry of molecules based on the repulsion between electron pairs.
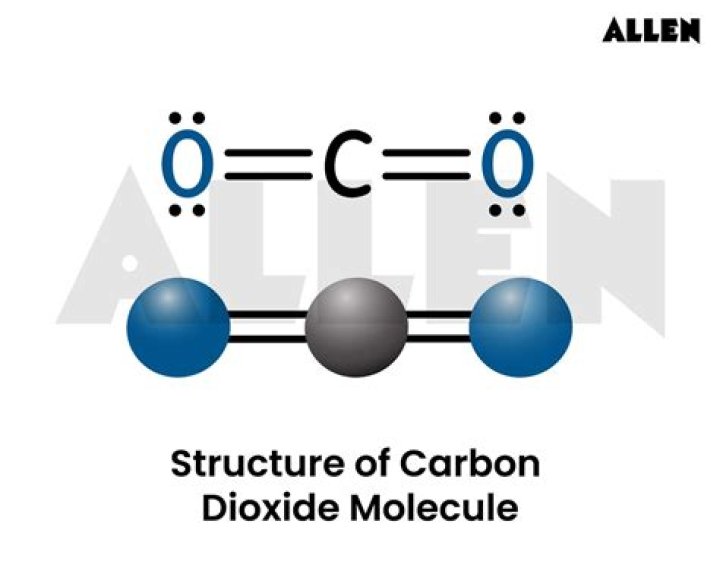
In the case of carbon dioxide, its molecular shape is linear, meaning the atoms are arranged in a straight line. This geometry results from the double bonds between carbon and oxygen atoms and the absence of lone pairs on the central carbon atom.
Carbon Dioxide Molecular Structure
Valence Electrons and Bonding
Carbon dioxide consists of one carbon atom and two oxygen atoms. Carbon has four valence electrons, while each oxygen atom has six valence electrons. To achieve a stable electron configuration, carbon forms double bonds with each oxygen atom, sharing two pairs of electrons. This arrangement satisfies the octet rule for all three atoms.
Double Bonds in CO2
- Carbon forms a double bond with each oxygen atom.
- Each double bond consists of one sigma (σ) bond and one pi (π) bond.
- The double bonds contribute to the molecule's stability and linear geometry.
Why Carbon Dioxide Has a Linear Geometry
The linear geometry of carbon dioxide arises from the arrangement of its atoms in space. The central carbon atom is bonded to two oxygen atoms, with no lone pairs of electrons present. According to VSEPR theory, the electron pairs in a double bond repel each other, forcing the atoms into a straight line. This results in a bond angle of 180 degrees, which is characteristic of linear molecules.
Bond Angle in Carbon Dioxide
The bond angle in carbon dioxide is precisely 180 degrees. This perfect alignment is due to the absence of lone pairs on the central carbon atom and the strong repulsion between the double bonds. The linear geometry ensures that the molecule is symmetrical, which has important implications for its physical and chemical properties.
Is Carbon Dioxide Polar or Nonpolar?
Despite having polar covalent bonds, carbon dioxide is a nonpolar molecule. The linear shape of CO2 causes the dipole moments of the C=O bonds to cancel each other out, resulting in a net dipole moment of zero. This nonpolarity makes carbon dioxide insoluble in water but highly soluble in organic solvents, which is important for its various applications.
Applications of Carbon Dioxide
Industrial Uses
- Carbonated beverages: CO2 is used to carbonate drinks, giving them their fizz.
- Fire extinguishers: CO2 is effective in extinguishing fires by displacing oxygen.
- Refrigeration: CO2 is used as a refrigerant in certain systems due to its non-toxic and non-flammable properties.
Environmental Uses
- Photosynthesis: CO2 is a key component in the process by which plants produce oxygen and glucose.
- Climate regulation: CO2 contributes to the greenhouse effect, helping maintain Earth's temperature.
Environmental Impact of Carbon Dioxide
While carbon dioxide is essential for life on Earth, excessive levels of CO2 in the atmosphere contribute to global warming and climate change. Human activities, particularly the burning of fossil fuels, have significantly increased atmospheric CO2 concentrations over the past century. This rise in CO2 levels leads to increased temperatures, melting polar ice caps, and rising sea levels, posing serious threats to ecosystems and human populations.
Factors Affecting Molecular Shape
Several factors influence the molecular shape of a compound, including:
- Number of bonding pairs and lone pairs of electrons.
- Type of bonds (single, double, or triple).
- Electronegativity of the atoms involved.
In the case of carbon dioxide, the absence of lone pairs and the presence of double bonds result in a linear molecular shape.
Conclusion and Call to Action
Carbon dioxide molecular shape is a fascinating subject that provides valuable insights into the properties and behavior of this essential molecule. Its linear geometry, bond angle, and nonpolar nature all contribute to its significance in both natural and industrial processes. Understanding these concepts is crucial for addressing environmental challenges and harnessing the potential of CO2 in various applications.
We encourage readers to explore further resources on this topic and share their thoughts in the comments section below. For more in-depth information, check out our other articles on chemistry and environmental science. Together, we can deepen our understanding of the world around us and work towards a sustainable future.
Data Sources and References:
- Chemistry: The Central Science by Brown, LeMay, Bursten, Murphy, Woodward, and Stoltzfus.
- Environmental Science by Miller and Spoolman.
- International Panel on Climate Change (IPCC) Reports.



