Understanding CO2 Polarity: A Comprehensive Guide
CO2 polarity is a fascinating subject that delves into the molecular structure and behavior of carbon dioxide. This gas plays a crucial role in our atmosphere and is essential for life on Earth. Understanding its properties, including its polarity, is vital for various scientific applications and environmental studies.
Carbon dioxide (CO2) is a naturally occurring gas that contributes to the greenhouse effect, which helps regulate the Earth's temperature. However, excessive CO2 levels can lead to global warming, making it imperative to study its properties in detail.
In this article, we will explore the concept of CO2 polarity, its molecular structure, and its implications in various fields. Whether you're a student, researcher, or simply curious about the science behind CO2, this guide will provide you with a thorough understanding of the topic.
Molecular Structure of CO2
Carbon dioxide (CO2) consists of one carbon atom and two oxygen atoms. The molecule has a linear structure, with the carbon atom bonded to each oxygen atom through double bonds. This linear arrangement plays a significant role in determining the polarity of CO2.
The molecular geometry of CO2 can be explained using the Valence Shell Electron Pair Repulsion (VSEPR) theory. According to this theory, the electron pairs around the central carbon atom repel each other, resulting in a linear shape with bond angles of 180 degrees.
Key Points:
- CO2 has a linear molecular structure.
- It consists of one carbon atom and two oxygen atoms.
- The double bonds between carbon and oxygen atoms contribute to its stability.
Understanding the Linear Shape
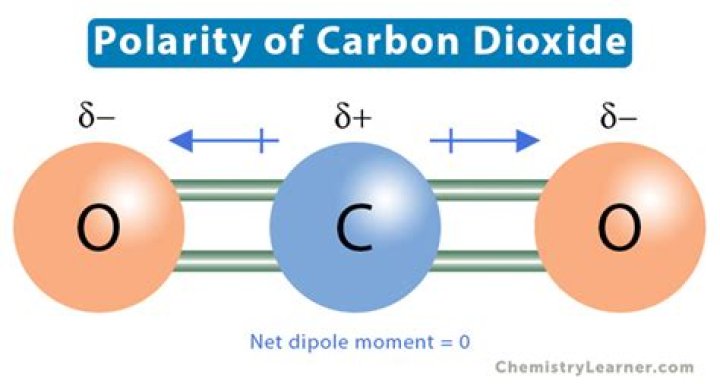
The linear shape of CO2 ensures that the molecule is symmetrical. This symmetry is crucial in determining the polarity of the molecule. In a linear molecule, the dipole moments of the bonds cancel each other out, resulting in a nonpolar molecule.
Is CO2 Polar or Nonpolar?
The question of CO2 polarity is often debated in chemistry classes. Despite having polar bonds, CO2 is considered a nonpolar molecule. This is due to the symmetrical arrangement of its atoms, which causes the dipole moments to cancel out.
Data: According to the Pauling scale, the electronegativity difference between carbon (2.55) and oxygen (3.44) is significant, indicating polar bonds. However, the linear geometry of CO2 ensures that the molecule as a whole is nonpolar.
Key Points:
- CO2 has polar bonds but is a nonpolar molecule.
- The symmetrical structure cancels out the dipole moments.
Why Symmetry Matters
Symmetry is a critical factor in determining the polarity of molecules. In the case of CO2, the linear arrangement ensures that the molecule is nonpolar, despite having polar bonds. This phenomenon is observed in other molecules with similar geometries, such as BeCl2 and CCl4.
Properties of CO2
Beyond its polarity, CO2 exhibits several unique properties that make it an important molecule in various fields. These properties include:
- Boiling point: -78.5°C
- Melting point: -56.6°C
- Molecular weight: 44.01 g/mol
- Solubility: CO2 is soluble in water, forming carbonic acid (H2CO3).
These properties make CO2 useful in industries such as food and beverage, fire extinguishers, and refrigeration.
CO2 Solubility in Water
When CO2 dissolves in water, it forms carbonic acid, a weak acid that contributes to the acidity of rainwater. This process is essential in maintaining the pH balance of natural water systems.
Applications of CO2
CO2 has numerous applications across various industries. Some of the most notable uses include:
- Carbonation of beverages: CO2 is used to add fizz to soft drinks and beer.
- Fire extinguishers: CO2 is effective in extinguishing fires by displacing oxygen.
- Refrigeration: CO2 is used as a refrigerant in certain applications.
- Greenhouse gas: CO2 is a key component of the Earth's atmosphere, regulating temperature.
These applications highlight the versatility and importance of CO2 in modern society.
CO2 in Agriculture
In agriculture, CO2 is used to enhance plant growth through controlled environments such as greenhouses. Elevated levels of CO2 can increase photosynthesis rates, leading to higher crop yields.
Environmental Impact of CO2
While CO2 is essential for life, excessive levels in the atmosphere contribute to global warming. The burning of fossil fuels and deforestation are major sources of CO2 emissions. Understanding the environmental impact of CO2 is crucial for addressing climate change.
Statistics: According to the Intergovernmental Panel on Climate Change (IPCC), CO2 levels have increased by 47% since the pre-industrial era. This rise is primarily due to human activities.
Key Points:
- CO2 is a major contributor to global warming.
- Reducing emissions is essential for combating climate change.
Solutions to Reduce CO2 Emissions
Efforts to reduce CO2 emissions include transitioning to renewable energy sources, improving energy efficiency, and reforestation. These solutions aim to mitigate the environmental impact of CO2 and promote sustainable development.
CO2 Bonding and Geometry
The bonding in CO2 involves double bonds between the carbon and oxygen atoms. These double bonds consist of one sigma bond and one pi bond. The geometry of CO2 is determined by the arrangement of electron pairs around the central carbon atom.
Key Points:
- CO2 has double bonds between carbon and oxygen atoms.
- The linear geometry results from the repulsion of electron pairs.
Significance of Double Bonds
The presence of double bonds in CO2 contributes to its stability and chemical properties. These bonds are stronger than single bonds, making CO2 a relatively inert molecule under normal conditions.
Dipole Moment of CO2
The dipole moment of a molecule is a measure of its polarity. In the case of CO2, the dipole moments of the individual bonds cancel each other out due to the linear geometry. This results in a net dipole moment of zero, confirming that CO2 is a nonpolar molecule.
Data: The dipole moment of CO2 is experimentally determined to be zero, supporting the theoretical predictions based on its molecular geometry.
Comparison with Other Molecules
CO2's dipole moment can be compared to other molecules, such as water (H2O) and ammonia (NH3). Unlike CO2, these molecules have nonzero dipole moments due to their asymmetrical shapes.
CO2 Polarity Compared to Other Molecules
When comparing the polarity of CO2 with other molecules, it becomes evident that molecular geometry plays a crucial role. For example, water (H2O) has a bent shape, resulting in a nonzero dipole moment and making it a polar molecule. In contrast, CO2's linear geometry ensures that it is nonpolar.
Key Points:
- CO2 is nonpolar due to its linear geometry.
- Water and ammonia are polar molecules due to their asymmetrical shapes.
Implications for Chemical Reactions
The polarity of a molecule influences its chemical behavior and reactivity. Nonpolar molecules like CO2 tend to interact differently with polar molecules, affecting their solubility and reactivity in various solvents.
Recent Research on CO2 Polarity
Recent studies on CO2 polarity focus on its implications in environmental science and industrial applications. Researchers are exploring ways to capture and utilize CO2 more effectively, reducing its environmental impact while harnessing its potential benefits.
Source: A study published in the Journal of Physical Chemistry highlights advancements in CO2 capture technology, emphasizing the importance of understanding its molecular properties.
Future Directions
Future research on CO2 polarity may lead to innovations in carbon capture and utilization. By understanding the molecular behavior of CO2, scientists can develop more efficient methods for reducing greenhouse gas emissions and promoting sustainability.
Conclusion
In conclusion, CO2 polarity is a complex yet fascinating topic that has significant implications in various fields. Despite having polar bonds, CO2 is a nonpolar molecule due to its symmetrical linear geometry. Understanding its properties and applications is essential for addressing environmental challenges and advancing scientific knowledge.
Call to Action: We encourage readers to explore further resources on CO2 and its role in our environment. Share this article with others to promote awareness and engage in discussions about climate change and sustainability.



