Understanding the Molar Mass of CaCO3: A Comprehensive Guide
When discussing chemical compounds, the molar mass of CaCO3 plays a significant role in various scientific and industrial applications. Calcium carbonate (CaCO3) is a widely used compound in fields such as construction, medicine, and environmental science. Understanding its molar mass is essential for anyone working with this compound.
Calcium carbonate is one of the most abundant minerals on Earth, found in limestone, marble, and chalk. Its chemical properties make it highly versatile, and its molar mass is a key factor in determining its behavior in different reactions. This article delves into the concept of molar mass, its calculation, and the significance of CaCO3 in various industries.
This guide aims to provide a detailed understanding of the molar mass of CaCO3, including its importance in chemical reactions, industrial applications, and environmental processes. Whether you're a student, researcher, or professional, this article will equip you with the knowledge needed to work effectively with calcium carbonate.
What is Molar Mass?
The molar mass of a substance is the mass of one mole of that substance, expressed in grams per mole (g/mol). It is derived from the atomic masses of the elements that make up the compound. For calcium carbonate (CaCO3), the molar mass is crucial in understanding its chemical and physical properties.
This concept is fundamental in chemistry as it allows scientists to calculate the amount of substance needed for specific reactions. The molar mass of CaCO3, for instance, is approximately 100.09 g/mol, which is determined by summing the atomic masses of calcium, carbon, and oxygen in the compound.
Overview of Calcium Carbonate (CaCO3)
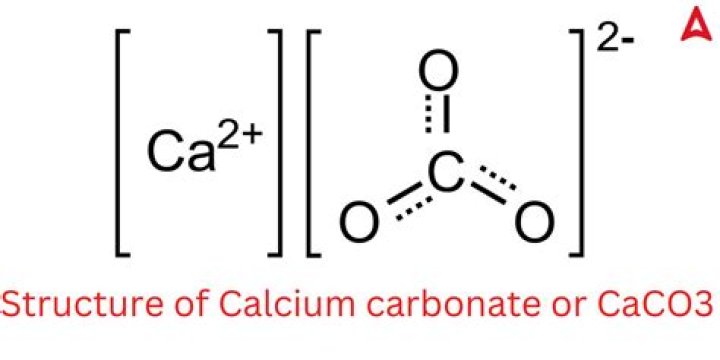
Calcium carbonate (CaCO3) is a chemical compound made up of calcium, carbon, and oxygen. It occurs naturally in minerals such as limestone, marble, and chalk. CaCO3 is a versatile compound with applications in construction, agriculture, and medicine.
Properties of Calcium Carbonate
CaCO3 is a white, crystalline powder that is insoluble in water. It reacts with acids to release carbon dioxide gas, making it useful in various industrial processes. Its molar mass is a critical factor in determining its reactivity and usability in different applications.
- Chemical Formula: CaCO3
- Molar Mass: 100.09 g/mol
- Appearance: White crystalline powder
- Solubility: Insoluble in water
Calculating the Molar Mass of CaCO3
To calculate the molar mass of CaCO3, you need to sum the atomic masses of its constituent elements. Calcium (Ca) has an atomic mass of approximately 40.08 g/mol, carbon (C) has an atomic mass of 12.01 g/mol, and oxygen (O) has an atomic mass of 16.00 g/mol. Since there are three oxygen atoms in CaCO3, the total molar mass is:
Molar Mass = (40.08) + (12.01) + (3 × 16.00) = 100.09 g/mol
This calculation is essential for understanding how CaCO3 behaves in chemical reactions and industrial processes.
Importance of Molar Mass in Chemistry
The molar mass of CaCO3 is vital in chemistry for several reasons:
- It allows chemists to determine the amount of substance needed for reactions.
- It helps in calculating the concentration of solutions.
- It provides insight into the physical and chemical properties of the compound.
Understanding the molar mass of CaCO3 enables scientists to predict its behavior in various environments, from laboratory settings to industrial applications.
Industrial Applications of CaCO3
Calcium carbonate is widely used in industries due to its versatile properties. Some of its key applications include:
Construction
CaCO3 is a primary component in cement and concrete production. Its molar mass affects its reactivity with other compounds, influencing the strength and durability of construction materials.
Pharmaceuticals
Calcium carbonate is used as a calcium supplement in medications and as an antacid to neutralize stomach acid. The molar mass of CaCO3 determines its effectiveness in these applications.
Paper Industry
CaCO3 is used as a filler in paper production, improving the paper's brightness and printability. Its molar mass affects its compatibility with other materials in the paper-making process.
Environmental Impact of Calcium Carbonate
Calcium carbonate plays a significant role in environmental processes. It is a major component of marine sediments and is involved in the carbon cycle. The molar mass of CaCO3 influences its behavior in natural environments:
- CaCO3 helps regulate ocean acidity by neutralizing carbon dioxide.
- It contributes to the formation of coral reefs and other marine structures.
- Its molar mass affects its solubility and reactivity in water bodies.
Understanding the environmental impact of CaCO3 is crucial for addressing issues such as ocean acidification and climate change.
Health Uses of CaCO3
Calcium carbonate is commonly used in healthcare for its health benefits:
- It serves as a dietary supplement to improve bone health.
- It is used as an antacid to relieve heartburn and indigestion.
- Its molar mass ensures precise dosing in medications, enhancing safety and efficacy.
Research from the National Institutes of Health (NIH) supports the use of CaCO3 in these applications, highlighting its importance in public health.
Common Questions About Molar Mass of CaCO3
What is the Molar Mass of CaCO3?
The molar mass of CaCO3 is approximately 100.09 g/mol, calculated by summing the atomic masses of calcium, carbon, and oxygen.
Why is Molar Mass Important?
Molar mass is crucial for determining the amount of substance needed for reactions, calculating solution concentrations, and understanding chemical properties.
How is CaCO3 Used in Medicine?
Calcium carbonate is used as a calcium supplement and antacid. Its molar mass ensures accurate dosing, making it safe and effective for medical use.
Measurement Techniques for Molar Mass
Several techniques are used to measure the molar mass of compounds like CaCO3:
- Mass Spectrometry: Provides precise measurements of molar mass.
- Titration: Determines the molar mass through chemical reactions.
- Gravimetric Analysis: Measures the mass of a substance before and after a reaction.
These techniques ensure accurate determination of molar mass, which is essential for scientific and industrial applications.
Future Perspectives on CaCO3 Research
Research on calcium carbonate continues to evolve, focusing on its applications in sustainable development and environmental protection:
- Developing eco-friendly CaCO3-based materials for construction.
- Exploring its role in carbon capture and storage technologies.
- Investigating new medical applications of CaCO3.
As technology advances, the importance of understanding the molar mass of CaCO3 will only increase, driving innovation in various fields.
Conclusion
In conclusion, the molar mass of CaCO3 is a fundamental concept in chemistry with wide-ranging applications. From construction to healthcare, calcium carbonate plays a vital role in modern industries. Understanding its molar mass enables scientists and professionals to harness its properties effectively.
We invite you to share your thoughts and questions in the comments section below. For more informative articles, explore our website and stay updated on the latest developments in science and technology.