Understanding the Molecular Model of CO2: A Comprehensive Guide
Carbon dioxide (CO2) is a fascinating molecule that plays a vital role in our environment, biology, and industrial processes. Its molecular structure is simple yet powerful, influencing everything from global climate to cellular respiration. Understanding the molecular model of CO2 is essential for anyone interested in chemistry, environmental science, or engineering.
This molecule, composed of one carbon atom and two oxygen atoms, exhibits unique properties that make it indispensable in various applications. From its linear geometry to its role as a greenhouse gas, CO2 is a cornerstone of modern science. In this article, we will delve deep into the molecular model of CO2, exploring its structure, properties, and significance in detail.
By the end of this guide, you will have a comprehensive understanding of CO2's molecular makeup, its importance in scientific research, and its real-world applications. Let’s dive in and explore the fascinating world of carbon dioxide!
Introduction to CO2 Molecular Model
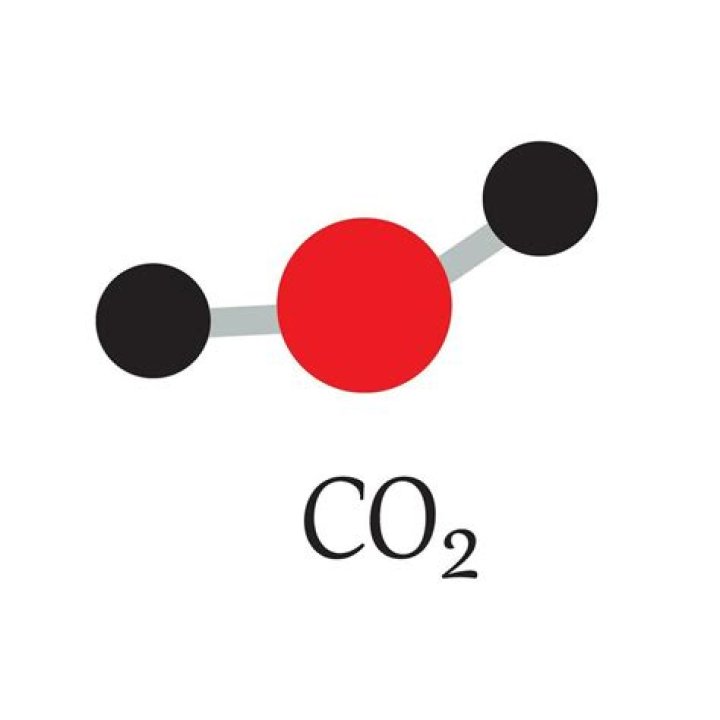
The molecular model of CO2 is a fundamental concept in chemistry that explains how carbon dioxide is structured at the atomic level. CO2 is a linear molecule consisting of one carbon atom bonded to two oxygen atoms through double bonds. This structure gives CO2 its unique properties, making it one of the most studied molecules in science.
Understanding the molecular model of CO2 is crucial for various fields, including atmospheric science, biochemistry, and materials engineering. Its simplicity belies its complexity, as the molecule exhibits fascinating behaviors under different conditions. For example, CO2 can exist as a gas, liquid, or solid (dry ice), depending on temperature and pressure.
Scientists use the molecular model of CO2 to predict its behavior in different environments. This knowledge is essential for addressing global challenges such as climate change, carbon capture, and renewable energy development. By studying the molecular model of CO2, researchers can design innovative solutions to mitigate its environmental impact while harnessing its potential for beneficial applications.
Structure of CO2 Molecule
Atomic Composition
The CO2 molecule consists of one carbon atom and two oxygen atoms. The carbon atom forms double bonds with each oxygen atom, resulting in a linear structure. This arrangement is due to the sp hybridization of the carbon atom, which allows for the formation of strong sigma and pi bonds with the oxygen atoms.
- Carbon atom: Located at the center of the molecule.
- Oxygen atoms: Positioned symmetrically on either side of the carbon atom.
Bond Length and Angle
The bond length between carbon and oxygen in CO2 is approximately 1.16 Å (angstroms). The molecule exhibits a bond angle of 180 degrees, confirming its linear geometry. This linear arrangement contributes to the molecule's nonpolar nature, as the dipole moments cancel each other out.
These structural features make CO2 highly stable and resistant to chemical reactions under normal conditions. However, under extreme conditions such as high temperature or pressure, CO2 can participate in various chemical processes, including decomposition and reaction with other molecules.
Properties of CO2
The molecular model of CO2 determines its physical and chemical properties. Some of the key properties of CO2 include:
- Nonpolar molecule: Due to its linear geometry and symmetrical distribution of charge, CO2 is nonpolar.
- High stability: CO2 is chemically stable under normal conditions, making it ideal for long-term storage and transportation.
- Low solubility in water: Although CO2 is slightly soluble in water, forming carbonic acid (H2CO3), its solubility is relatively low compared to other gases.
- Greenhouse gas: CO2 is a potent greenhouse gas, contributing significantly to global warming and climate change.
Understanding these properties is essential for designing technologies that can capture, store, or utilize CO2 effectively. For example, researchers are exploring methods to convert CO2 into valuable products such as fuels, chemicals, and building materials.
Bonding in CO2
Double Bonds
The bonding in CO2 involves double bonds between the carbon and oxygen atoms. Each double bond consists of one sigma bond and one pi bond. The sigma bond is formed by the head-on overlap of sp hybridized orbitals, while the pi bond is formed by the side-to-side overlap of p orbitals.
This bonding arrangement gives CO2 its exceptional stability and resistance to chemical reactions. The high bond energy of the double bonds makes it difficult for CO2 to break apart under normal conditions.
Resonance Structures
CO2 exhibits resonance, meaning that its bonding can be represented by multiple equivalent Lewis structures. These resonance structures show the delocalization of electrons between the carbon and oxygen atoms, further enhancing the molecule's stability.
Resonance plays a critical role in determining the reactivity and properties of CO2. For example, the delocalization of electrons reduces the likelihood of CO2 participating in chemical reactions, making it an ideal molecule for long-term storage and transportation.
Molecular Geometry of CO2
The molecular geometry of CO2 is linear, with a bond angle of 180 degrees. This geometry arises from the sp hybridization of the carbon atom, which results in a linear arrangement of the sigma bonds. The pi bonds formed by the p orbitals are perpendicular to the molecular plane, further stabilizing the molecule.
The linear geometry of CO2 contributes to its nonpolar nature, as the dipole moments of the C=O bonds cancel each other out. This makes CO2 an excellent solvent for nonpolar substances and a key component in various industrial processes.
Understanding the molecular geometry of CO2 is essential for predicting its behavior in different environments. For example, the linear shape of CO2 allows it to interact with other molecules in specific ways, influencing its reactivity and solubility.
Applications of CO2
Industrial Uses
CO2 has numerous industrial applications, including:
- Carbonation of beverages: CO2 is used to carbonate soft drinks, beer, and other beverages, giving them their characteristic fizz.
- Fire extinguishers: CO2 is an effective fire suppressant, as it displaces oxygen and smothers flames.
- Refrigeration: CO2 is used as a refrigerant in certain applications, particularly in food processing and transportation.
Medical Applications
In the medical field, CO2 is used for:
- Endoscopic procedures: CO2 is used to inflate body cavities during endoscopic surgeries, providing better visibility for surgeons.
- Laser treatments: CO2 lasers are widely used in dermatology and other medical specialties for precise cutting and coagulation.
These applications highlight the versatility and importance of CO2 in modern industry and healthcare.
CO2 and the Environment
CO2 is a major contributor to global warming and climate change. As a greenhouse gas, it traps heat in the Earth's atmosphere, leading to rising temperatures and altered weather patterns. The increasing levels of CO2 in the atmosphere are primarily due to human activities such as fossil fuel combustion, deforestation, and industrial processes.
To mitigate the environmental impact of CO2, scientists are developing technologies for carbon capture and storage (CCS). These technologies aim to capture CO2 emissions from power plants and industrial facilities, storing them underground or converting them into useful products.
Additionally, efforts are underway to reduce CO2 emissions through renewable energy adoption, energy efficiency improvements, and sustainable practices. By addressing the environmental challenges posed by CO2, we can work towards a more sustainable future.
Biological Role of CO2
In biological systems, CO2 plays a crucial role in cellular respiration and photosynthesis. During cellular respiration, CO2 is produced as a byproduct of energy production in cells. This CO2 is then transported to the lungs, where it is expelled from the body during exhalation.
In photosynthesis, CO2 is absorbed by plants and converted into glucose and oxygen using sunlight. This process is essential for maintaining the balance of CO2 in the atmosphere and supporting life on Earth.
Understanding the biological role of CO2 is vital for addressing global challenges such as food security, climate change, and environmental sustainability.
Industrial Uses of CO2
Beyond its environmental and biological significance, CO2 has numerous industrial applications. These include:
- Enhanced oil recovery: CO2 is injected into oil reservoirs to increase oil production by reducing viscosity and improving flow.
- Chemical synthesis: CO2 is used as a reactant in the production of various chemicals, including urea, methanol, and polycarbonates.
- Food processing: CO2 is used for chilling and freezing foods, as well as for modified atmosphere packaging to extend shelf life.
These applications demonstrate the versatility and importance of CO2 in modern industry. By harnessing the unique properties of CO2, we can develop innovative solutions to address global challenges.
Future Prospects of CO2 Research
The study of CO2 is an active area of research, with scientists exploring new ways to capture, store, and utilize this molecule. Some promising areas of research include:
- Carbon capture and utilization (CCU): Developing technologies to convert CO2 into valuable products such as fuels, chemicals, and building materials.
- Artificial photosynthesis: Mimicking the natural process of photosynthesis to produce energy and chemicals from CO2 and sunlight.
- Advanced materials: Designing new materials that can efficiently capture and store CO2, such as metal-organic frameworks (MOFs) and porous carbon materials.
By advancing our understanding of CO2 and its applications, we can create a more sustainable future for generations to come.
Kesimpulan
The molecular model of CO2 is a fundamental concept in chemistry that explains the structure, properties, and behavior of this important molecule. From its linear geometry to its role as a greenhouse gas, CO2 plays a vital role in our environment, biology, and industry. Understanding the molecular model of CO2 is essential for addressing global challenges such as climate change, energy production, and environmental sustainability.
We encourage readers to explore further resources on CO2 and its applications. By sharing this article and engaging in discussions, you can contribute to the growing body of knowledge on this fascinating molecule. Together, we can work towards a more sustainable and prosperous future for all.