Unlocking the Mystery: One End of a Battery NYT Answer
Have you ever pondered the significance of "one end of a battery" in the context of the New York Times puzzles? This phrase has intrigued many puzzle enthusiasts and sparked curiosity among word game lovers worldwide. The New York Times crossword puzzle, a beloved pastime for millions, often includes clever clues that require both knowledge and creativity to solve. Today, we delve into the fascinating world of batteries and their connection to this iconic puzzle.
Understanding the concept of "one end of a battery" is not only about solving crossword puzzles. It's also about appreciating the intricate design and functionality of batteries, which power countless devices in our daily lives. By exploring this topic, we aim to provide clarity and insights that will enhance your puzzle-solving skills and broaden your knowledge.
Join us on this journey as we unravel the mystery behind "one end of a battery" and its relevance in the New York Times puzzle. Whether you're a seasoned puzzle enthusiast or a curious beginner, this article promises to deliver valuable information and engaging content.
Introduction to One End of a Battery NYT Answer
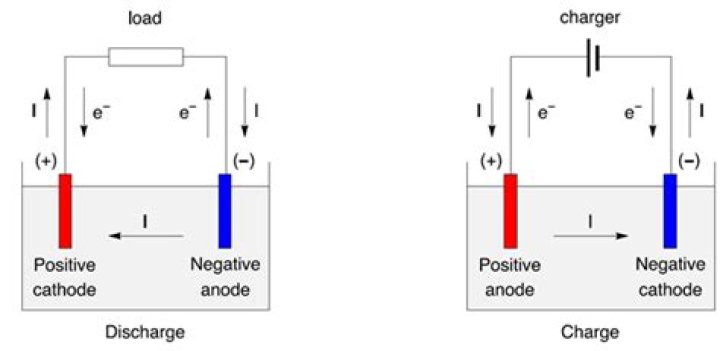
The phrase "one end of a battery" often appears in the New York Times crossword puzzle, challenging solvers to think critically about the components of a battery. At its core, a battery is a device that stores chemical energy and converts it into electrical energy. Understanding the ends of a battery—positive and negative terminals—is fundamental to solving this particular crossword clue.
In crossword puzzles, the answer to "one end of a battery" is typically "pole." This is because batteries have two poles: the positive pole and the negative pole. These poles play a crucial role in the flow of electric current, making them essential components of any battery-powered device.
Battery Basics: Understanding the Components
What Makes Up a Battery?
A battery consists of several key components, each with a specific function. The primary parts include:
- Electrodes: The positive and negative terminals where electric current enters or exits the battery.
- Electrolyte: A substance that facilitates the flow of ions between the electrodes.
- Separator: A material that prevents the electrodes from touching while allowing ions to pass through.
- Collector: A conductor that transfers electric current from the electrodes to external circuits.
Each of these components works together to generate and store electrical energy, making batteries indispensable in modern technology.
The Role of "One End of a Battery" in NYT Crossword
Why This Clue is Popular
The New York Times crossword puzzle is renowned for its clever and thought-provoking clues. "One end of a battery" is a classic example of a clue that requires solvers to think about the basic structure of a battery. The simplicity of the clue belies its complexity, as it demands an understanding of both language and science.
For crossword enthusiasts, solving this clue is a satisfying experience that combines knowledge with problem-solving skills. It also serves as a reminder of the importance of batteries in our daily lives, from powering remote controls to fueling electric vehicles.
Types of Batteries and Their Uses
Exploring Different Battery Technologies
Batteries come in various types, each designed for specific applications. Some of the most common types include:
- Alkaline Batteries: Widely used in household devices due to their affordability and reliability.
- Lithium-Ion Batteries: Known for their high energy density and used in smartphones, laptops, and electric cars.
- Nickel-Cadmium Batteries: Often used in power tools and medical equipment.
- Lead-Acid Batteries: Commonly found in automotive applications due to their durability and cost-effectiveness.
Understanding the differences between these battery types can help you choose the right one for your needs and appreciate the role of "one end of a battery" in powering modern technology.
Scientific Aspects of Battery Ends
The Science Behind Positive and Negative Poles
The positive and negative poles of a battery are critical to its operation. The positive pole, or cathode, attracts electrons, while the negative pole, or anode, releases them. This flow of electrons creates an electric current, which powers devices connected to the battery.
Scientists continue to study the properties of battery poles to improve efficiency and sustainability. Advances in materials science and electrochemistry are paving the way for more powerful and environmentally friendly batteries.
Historical Perspective of Batteries
From Voltaic Pile to Modern Batteries
The history of batteries dates back to the early 19th century when Italian physicist Alessandro Volta invented the voltaic pile, the first true battery. This groundbreaking invention laid the foundation for modern battery technology.
Over the years, scientists and engineers have made significant advancements in battery design and performance. Today, batteries are more efficient, reliable, and environmentally friendly than ever before, thanks to ongoing research and innovation.
Practical Applications of Battery Technology
How Batteries Power Our World
Batteries play a vital role in powering a wide range of devices and systems. Some of the most common applications include:
- Consumer Electronics: Smartphones, tablets, and laptops rely on rechargeable batteries for portability and convenience.
- Transportation: Electric vehicles and hybrid cars use advanced battery systems to reduce reliance on fossil fuels.
- Renewable Energy Storage: Solar and wind power systems utilize batteries to store energy for use during periods of low generation.
- Medical Devices: Pacemakers and other life-saving devices depend on batteries for uninterrupted operation.
As battery technology continues to evolve, its applications will expand, further enhancing our ability to harness and utilize energy efficiently.
Environmental Impact of Battery Disposal
Addressing the Challenges of Battery Waste
While batteries are essential to modern life, their disposal poses significant environmental challenges. Improperly discarded batteries can release toxic chemicals into the soil and water, harming ecosystems and human health.
To mitigate these effects, governments and organizations are promoting recycling programs and encouraging the development of eco-friendly battery technologies. By responsibly managing battery waste, we can reduce its environmental impact and protect the planet for future generations.
Future Trends in Battery Innovation
What Lies Ahead for Battery Technology
The future of battery technology is bright, with researchers exploring new materials and designs to enhance performance and sustainability. Some promising developments include:
- Solid-State Batteries: Offering higher energy density and improved safety compared to traditional lithium-ion batteries.
- Graphene-Based Batteries: Providing faster charging times and longer lifespans.
- Sodium-Ion Batteries: A cost-effective alternative to lithium-ion batteries, using abundant sodium instead of scarce lithium.
As these innovations come to fruition, they will revolutionize the way we power our devices and contribute to a more sustainable energy future.
Conclusion and Final Thoughts
In conclusion, the phrase "one end of a battery" is more than just a crossword clue. It represents a fundamental aspect of battery technology that powers countless devices in our daily lives. By understanding the components, functions, and applications of batteries, we can appreciate their significance and contribute to their responsible use and development.
We encourage you to share your thoughts and insights in the comments section below. Are there other aspects of battery technology you'd like to explore? Let us know, and don't forget to check out our other articles for more fascinating content. Together, let's unlock the mysteries of modern science and technology!
Sources:

